簡介
離子強度是溶液中離子濃度的量度,是溶液中所有離子濃度的函式,定義如下:
 離子強度
離子強度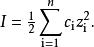 離子強度
離子強度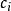 離子強度
離子強度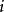 離子強度
離子強度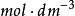 離子強度
離子強度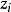 離子強度
離子強度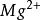 離子強度
離子強度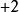 離子強度
離子強度其中:是離子的摩爾濃度(單位),是離子所帶的電荷數,如鎂離子就是。
離子化合物溶於水中時,會解離成離子。水溶液中電解質的濃度會影響到其他鹽類的溶解度。尤其是當易溶的鹽類溶於水中時,會大幅降低難溶鹽類的溶解度。而影響的強弱程度就稱為離子強度。
體積摩爾濃度
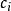 離子強度
離子強度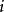 離子強度
離子強度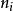 離子強度
離子強度體積摩爾濃度(英語: molarity,通常以大寫M表示)是化學的一種通用濃度單位,體積摩爾濃度定義為指構成溶液的某組分的物質的量除以溶液的體積V:
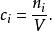 離子強度
離子強度在大多數情況下,體積摩爾濃度指溶質的體積摩爾濃度,即溶質的物質的量除以溶液的體積。
參見
•摩爾數
•濃度